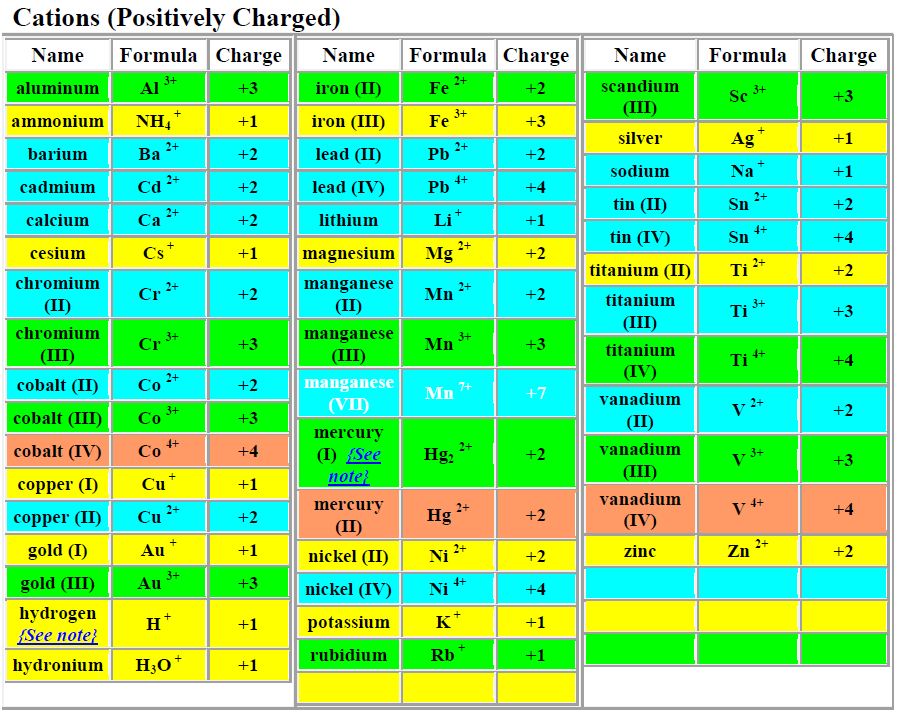 
The electric current generated by a battery arises because electrons are transferred from a reducing agent to an oxidizing agent through the external circuitry. Redox reactions are the source of the energy of batteries. 2Fe(s) + 2H 2O(l) + O 2(g) → 2Fe(OH) 2(s) Here iron metal is oxidized to iron dihydroxide (Fe(OH) 2) elemental oxygen (O 2) is the oxidizing agent. The reaction occurs with each magnesium atom giving up two electrons and being oxidized and each oxygen atom accepting two electrons and being reduced.Īnother common redox reaction is one step in the rusting of iron in damp air. The product is an ionic compound, made up of Mg 2+ and O 2− ions. For example, magnesium burns in oxygen to form magnesium oxide (MgO). Simple redox reactions include the reactions of an element with oxygen. This has the effect of reducing the real or apparent electric charge on an atom in the substance being reduced and of increasing the real or apparent electric charge on an atom in the substance being oxidized. Oxidation-reduction (redox) reactions involve the transfer of one or more electrons from a reducing agent to an oxidizing agent. In contrast, reactions of acids with bases in water involve proton (H +) transfer from an acid to a base. Oxidation-reduction reactions occur with electron transfer between reagents. Two types of reactions involve transfer of a charged species. In many cases the properties of a mixture are determined primarily by forces that are more properly classified as chemical rather than as. SpaceNext50 Britannica presents SpaceNext50, From the race to the Moon to space stewardship, we explore a wide range of subjects that feed our curiosity about space!.Learn about the major environmental problems facing our planet and what can be done about them! Saving Earth Britannica Presents Earth’s To-Do List for the 21st Century.Britannica Beyond We’ve created a new place where questions are at the center of learning. 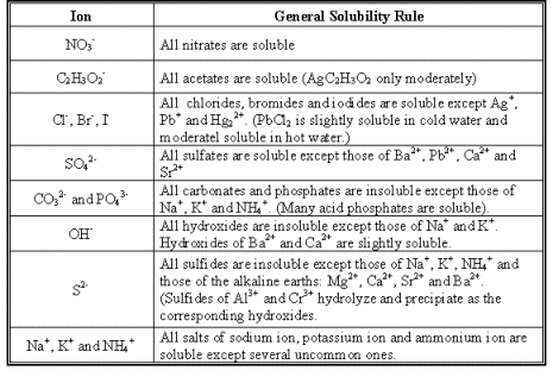
100 Women Britannica celebrates the centennial of the Nineteenth Amendment, highlighting suffragists and history-making politicians.COVID-19 Portal While this global health crisis continues to evolve, it can be useful to look to past pandemics to better understand how to respond today.Student Portal Britannica is the ultimate student resource for key school subjects like history, government, literature, and more.From tech to household and wellness products. Britannica Explains In these videos, Britannica explains a variety of topics and answers frequently asked questions.This Time in History In these videos, find out what happened this month (or any month!) in history.#WTFact Videos In #WTFact Britannica shares some of the most bizarre facts we can find.Demystified Videos In Demystified, Britannica has all the answers to your burning questions.Britannica Classics Check out these retro videos from Encyclopedia Britannica’s archives.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
